One-a-day anti-HIV drug OK’d
Fulfilling a long-held goal of AIDS researchers, federal regulators on Wednesday approved the first anti-HIV drug that requires patients to take only one pill a day.
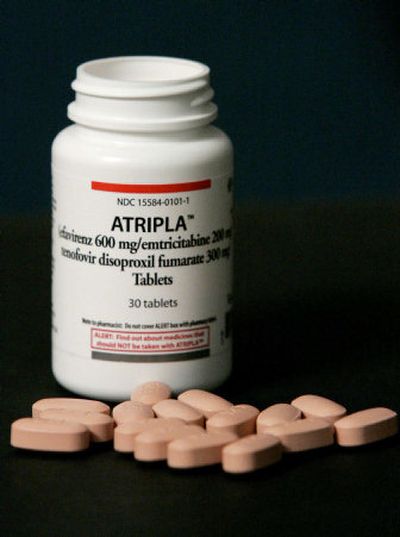
The new drug, called Atripla, combines the three most widely prescribed HIV drugs into one pill, providing patients with the simplest possible medication regimen.
The new drug could slow the development of resistance to the AIDS virus by increasing patients’ adherence to their drug regimens. It could also lower their out-of-pocket expenses by reducing the number of co-pays for their insurance.
The approval is “a landmark in the treatment of HIV/AIDS,” said Andrew C. von Eschenbach, acting commissioner of the Food and Drug Administration, in announcing the approval in Washington.
The development of Atripla represents the first time that two pharmaceutical manufacturers have combined their HIV drugs into one pill.
The salmon-colored pill, about the size of a multivitamin, contains Viread and Emtriva, produced by Gilead Sciences Inc. of Foster City, Calif., and Sustiva, manufactured by New York-based Bristol-Meyers Squibb Co.
The companies said that Atripla would be available to consumers within seven business days and that its wholesale price would be $1,150.88 for a 30-day supply, the same price as the drugs sold separately.
The three drugs, whose generic names are tenofovir, emtricitabine and efavirenz, respectively, are all reverse transcriptase inhibitors, which block a key enzyme that is unique to HIV. Reverse transcriptase inhibitors were the first family of HIV drugs developed and are still the most widely prescribed family of drugs for newly diagnosed patients.
When a patient develops resistance to one or more of the drugs, physicians typically add a second family of drugs called protease inhibitors. Experts said companies were already looking to find ways to incorporate them into the pill as well.
Sustiva and Emtriva are already marketed in a once-daily combination pill called Truvada, so many patients are already taking only two pills a day.
But studies of various types of drugs have shown that “the fewer pills you can get, the better the compliance,” said Dr. Joseph Cadden of the University of Southern California’s Keck School of Medicine. “That’s a key factor in AIDS, where you have to have an adherence of 95 percent plus” to prevent resistance developing.
The efforts of the two companies to produce the pill is especially important to HIV-positive people who remember the early days of HIV drug cocktails when patients took 30 or more pills daily.
“People were talking about once-a-day as a magical, wonderful, far-off moment,” said Craig E. Thompson, executive director of AIDS Project Los Angeles. “It was one of those things we were waiting for, and we’ve done a great job in getting there.”