Spokane cardiologist using new absorbable stents to treat patients with coronary artery disease
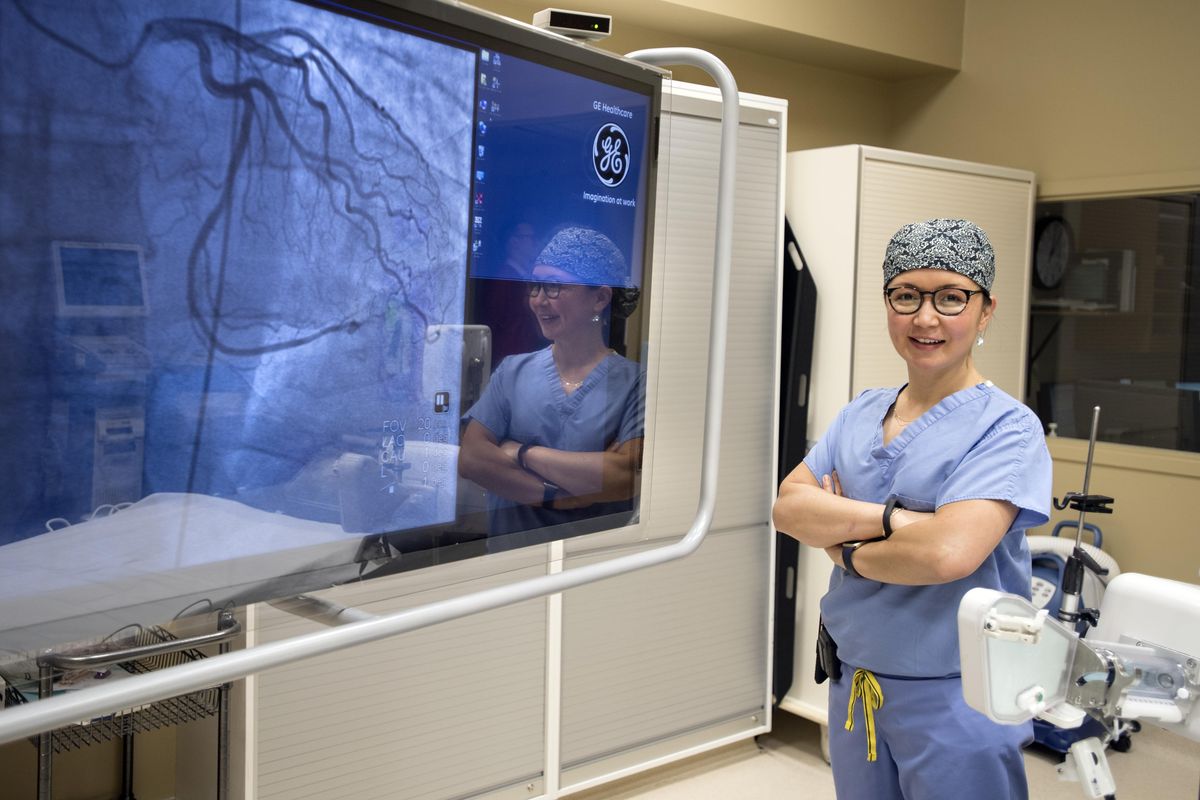
A cardiologist at Deaconess Hospital has become the first in Spokane to use a new device to treat coronary artery disease.
Dr. Ellie Mueller, an interventional cardiologist, is now using bioreabsorbable stents to re-open blocked arteries in some patients. The stents – essentially tiny polymer tubes, inserted into blocked arteries to increase blood flow near the heart – are gradually absorbed into the body over a three-year period.
Coronary artery disease is the most common type of heart disease, and the leading cause of death for Americans, according to the U.S. National Library of Medicine.
In patients with the disease, arteries feeding oxygen-rich blood to the heart become harder and narrower because of the buildup of cholesterol and plaque.
Traditionally, cardiologists like Mueller have used metal stents: tiny springlike devices a few millimeters wide that are inserted into blocked arteries, helping them stay open.
It’s a common procedure for cardiologists. Mueller estimates about one-third of her surgeries involve inserting stents.
Metal stents work well for most patients, but in some cases the stent itself ends up blocked, sometimes within a few years. Inflammation from the stent surgery can cause scarring, which contributes to blockage down the road.
“(The stent) narrows just like the artery,” Mueller said. “Some people scar more. Some people scar less. Some people scar very quickly, and they come back within a few years.”
The bioreabsorbable stent is made from several materials that can be reabsorbed into the body, chiefly lactic acid. When it’s reabsorbed, it’s easier for the artery to stay open, and for a new stent to be inserted if needed.
The bioreabsorbable stents are a viable alternative for patients who have already had metal stents inserted and had those stents become blocked from scarring.
“I don’t want to put another metal stent inside an already scarred stent,” Mueller said. “I want to put something in that dissolves and keeps the artery open.”
The new stent was approved by the FDA last fall.
A clinical trial presented at the American College of Cardiology in March found patients treated with the reabsorbable stent had slightly higher rates of major heart problems, including heart attacks or follow-up surgery after two years, than patients receiving a metal stent.
That data prompted the FDA to issue a warning reminding providers to monitor for cardiac symptoms and only use the reabsorbable stents in appropriately-sized vessels.
Mueller said those trials included a 2.25 millimeter stent that was responsible for most of the complications. After the trial results came out, the company began making stents sized only 2.5 millimeters and up, which cut down on the risk of complications.
While metal stents are usually the first line of defense for opening blocked arteries, the bioreabsorbable stent gives surgeons like Mueller another tool for patients with many blocked arteries, especially younger patients who are likely to need more intervention down the road.
“Coronary artery disease is a chronic condition,” Mueller said. “Once you have a blockage you will have a tendency to have more blockages down the road.”
Some patients may need 30 stents inserted, making new metal placements difficult.
“At some point I cannot fit any more metal in there,” she said.